|
Welcome to this issue of the CDER ARC Program Newsletter as we reflect on accomplishments of 2025 and look toward those in 2026. We are excited to share:
- ARC Program updates
-
2025 Novel Drug Approvals, Orphan Drug Designations, and Grant Updates
-
Ongoing News, Events, Engagement, and Resources
To get in touch, please email CDER_ARC_Program@fda.hhs.gov.
A Message from the ARC Program Team
2025 was an opportunity for the ARC program to affirm its purpose and make plans for the future. The ARC Program team has maintained focus on our three pillars of Scientific and Regulatory Innovation, Engagement and Education, and Operations that continue to guide all our ARC and collaboratively led initiatives. Our Program is the heart of a network of individuals and institutions working together toward improved treatments and cures for rare diseases. These collaborations are a catalyst for innovation.
-
30+ public speaking engagements
-
57+ workshops, consults, or meetings to facilitate rare disease drug development
-
45,000 rare disease news subscribers
-
Maintenance of FDA-TRACK’s dashboard, which can be filtered to highlight rare disease-specific cumulative approvals by quarter
-
40+ rare and orphan diseases represented within Critical Path Institute’s Rare Disease Cures Accelerator data-sharing and integration platform
Rare Disease Initiatives, News, and Opportunities
CDER Publishes 2025 Novel Drug Approvals Report
CDER released its Advancing Health Through Innovation: New Drug Therapy Approvals 2025 report summarizing the 46 novel drug approvals of the past calendar year. Seventy percent of drugs (32) were approved in the U.S. before approval in other countries, and exactly half (23) received orphan drug designation. Learn more about CDER’s novel drug approvals for 2025 on this webpage.
Office of Orphan Products Development (OOPD) 2025 Designations and Grants
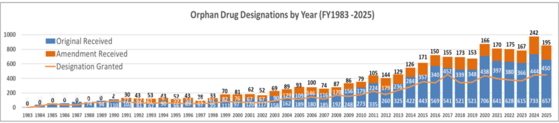
In FY25, the FDA Office of Orphan Products Development (OOPD) in the Office of the Chief Medical Officer, Office of the Commissioner, received 657 original orphan drug designation (ODD) requests and 195 amendments. A total of 450 designations were granted. Search orphan drug designations and approvals on this webpage.
 2025 Orphan Products Grants Awarded
Clinical trial grants for orphan products are a proven method to foster the development of new safe and effective medical products for rare diseases and conditions. Learn more at the OOPD Clinical Trial Grants Awarded webpage.
2025 Rare Neurodegenerative Disease Grants Awarded
Launched when the Accelerating Access to Critical Therapies for ALS (also referred to as ACT for ALS) law was enacted in December 2021, the Rare Neurodegenerative Disease grants program supports research in amyotrophic lateral sclerosis (ALS) and other rare neurodegenerative diseases in adults and children. Learn more about these OOPD grants at the OOPD RND Grants Awarded webpage. Learn more about ACT for ALS here.
Selected Rare Disease Approvals and Rare Pediatric Disease Priority Review Vouchers (RPD PRVs)
FDA approved Yuviwel (navepegritide) for injection on Feb. 27, 2026, to increase linear growth in pediatric patients aged 2 years and older with achondroplasia, the most common form of dwarfism, with open epiphyses. Yuviwel approval earned a RPD PRV.
FDA approved Zycubo (copper histidinate) injection as the first treatment for Menkes disease in pediatric patients. Approved on Jan. 12, 2026, this marked the first new molecular entity approval of 2026. Zycubo approval earned a RPD PRV.
On Nov. 3. 2025, FDA approved Kygevvi (doxecitine and doxribtimine) powder to treat thymidine kinase 2 deficiency (TK2d), a very rare mitochondrial disease in adults and pediatric patients who start to show symptoms when they are 12 years old or younger. This was the first approval to treat TK2d. Kygevvi approval earned a RPD PRV.
You can find other notable CDER rare disease approvals on the Rare Disease Approvals webpage, part of the recently updated ARC website.

Other News
ARC Participation in FDA Celebration of Rare Disease Day
The Rare Diseases Team’s Andrea Bell-Vlasov kicked off Rare Disease Day as the first presenter, discussing the Learning and Education to ADvance and Empower Rare Disease Drug Developers (LEADER 3D) program. During her presentation, she provided information on LEADER 3D’s purpose and progress since its inception, including the suite of educational materials available to rare disease drug developers on the LEADER 3D webpage.
FDA's Rare Disease Endpoint Advancement (RDEA) Pilot Program Accepting Proposals to Support Novel Endpoint Development for Rare Disease Treatments
RDEA seeks to advance rare disease drug development programs by providing a mechanism for sponsors to collaborate with FDA throughout the efficacy endpoint development process. The next quarterly submission deadline is June 30, 2026. Learn more here.
Patient Focused Drug Development (PFDD) Meetings and Patient Listening Sessions for Rare Diseases
To support the patient community, patient advocates, researchers, drug developers, and federal agencies, FDA provides summaries of Patient Listening Sessions and PFDD Meeting Reports on its website. To find out more about or request a Patient Listening Sessions, please visit FDA Patient Listening Sessions | FDA. More information about PFDD Meetings can be found at FDA-led Patient-Focused Drug Development (PFDD) Public Meetings | FDA. To request a PFDD meeting please see Externally led Patient-Focused Drug Development Meetings | FDA, and to view upcoming externally led meetings, see this webpage.
FDA’s Rare Disease Innovation Hub Releases 2026 Strategic Agenda
On Feb. 2, 2026, the FDA Rare Disease Innovation Hub released its second annual Strategic Agenda, publicly communicating its goals and priorities for the upcoming year to the rare disease community. The Strategic Agenda reflects extensive input from and partnership with the larger rare disease community. For more information, visit the Hub’s webpage.
|
Mark Your Calendars
FDA to Hold Workshop on Advancing Novel Surrogate Endpoints for Rare Disease Drug Development
FDA is convening a one-day virtual public workshop on May 18, 2026, focused on the development of surrogate endpoints for drugs and biological products intended to treat rare diseases. The workshop will discuss mechanistic and translational evidence considerations for supporting a novel surrogate endpoint in rare disease marketing applications, including a case study highlighting the evidentiary package to support the use of a novel surrogate endpoint. FDA, clinicians, researchers, and other parties will also discuss various relevant and emerging translational science topics to support the development of novel surrogate endpoints, such as new approach methodologies, digital health technology, and patient-level real-world data.
In Case You Missed It: Recent Events & Programming
C3TI Public Workshop on Clinical Trial Innovation
This hybrid workshop on April 14, 2026, explored progress in clinical trial innovation to improve the efficiency of drug development. Presenters discussed lessons learned from recent efforts, the benefits of adopting innovative approaches in clinical trials, and persistent challenges. Participants discussed strategies to bridge from policy to implementation to broader adoption of innovative approaches in the design and conduct of clinical trials
RISE Workshop on Data Sharing
This public workshop, co-convened by the Duke-Margolis Institute for Health Policy and the FDA Rare Disease Innovation Hub, was designed for all stakeholders in the rare disease community to explore data sharing as it pertains to informing development and regulatory review for rare disease therapies. To learn more, visit the workshop site. To share ideas for future RISE Workshops, submit a proposal to the Rare Disease Innovation Hub’s 2026 public docket.
From Vision to Reality: 6 Years of RDCA-DAP, Driving Drug Development Solutions and Innovations in Data Sharing
C-Path's Rare Disease Cures Accelerator-Data and Analytics Platform (RDCA-DAP) team held a workshop, From Vision to Reality: 6 Years of RDCA-DAP, Driving Drug Development Solutions and Innovations in Data Sharing, on Dec. 11, 2025. The team shared how they promote data sharing to create a growing database in rare disease, fostering a collaborative data ecosystem with partnering platforms and facilitating drug development activities.
ADEPT 10: Addressing Challenges in Neonatal Product Development – Leveraging Rare Disease Frameworks
On Dec. 10-11, 2025, FDA held a public workshop, "Advancing the Development of Pediatric Therapeutics (ADEPT) 10: Addressing Challenges in Neonatal Product Development – Leveraging Rare Disease Frameworks. The aim of the public workshop was to discuss common challenges in neonatal and rare disease product development and identify opportunities to leverage rare disease product development frameworks in the neonatal product development space.
Individualized Therapies on the RISE
This public workshop, co-convened by the Rare Disease Innovation Hub at the FDA and the Duke-Margolis Institute for Health Policy and held Nov. 25, 2025, examined the emerging science and regulatory environment for individualized medicine programs, including nonclinical data recommendations, clinical assessments, regulatory submission structure, and additional information necessary to support the development, evaluation, and potential commercialization of these treatments. For workshop materials and recording, visit Individualized Therapies on the RISE.
Rare Disease Resources
|